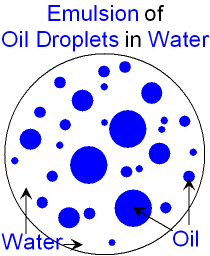
The addition of ethylene oxide increases water solubility, propylene oxide decreases it.Ĭommercially available demulsifier formulations are typically a mixture of two to four different chemistries, in carrier solvent(s) such as xylene, heavy aromatic naphtha (HAN), Isopropanol, methanol, 2-Ethylhexanol or diesel. Simple emulsions are either oil suspended in an aqueous phase (o/w), or water suspended in oil (w/o). The above are usually ethoxylated (and/or propoxylated) to provide the desired degree of water/oil solubility. The word emulsion comes from the Latin word meaning 'to milk' (milk is one example of an emulsion of fat and water). In other words, an emulsion is a special type of mixture made by combining two liquids that normally don't mix. Oil and the mixtures of water are emulsions when. An emulsion is a colloid of two or more immiscible liquids where one liquid contains a dispersion of the other liquids. Base catalysed phenol-formaldehyde resins In simple words, emulsions are colloids in which both the dispersed phase and dispersion medium are liquids.Emulsions consist of two parts: the dispersed phase and the. Emulsions are colloids, which are homogeneous mixtures consisting of particles larger than molecules that scatter light, but are small enough that they don’t separate. Acid catalysed phenol-formaldehyde resins emulsion, in physical chemistry, mixture of two or more liquids in which one is present as droplets, of microscopic or ultramicroscopic size, distributed throughout the other. An emulsion is defined as a mixture of two or more normally immiscible (unmixable) liquids.If the majority of the water and salt are not removed, significant corrosion problems can occur in the refining process.ĭemulsifiers are typically based on the following chemistry: This water (and salt) must be removed from the crude oil prior to refining. Word forms: (regular plural) oil-in-water emulsions. They are commonly used in the processing of crude oil, which is typically produced along with significant quantities of saline water. This isnt to say that all colloids are emulsions. Despite the fact that emulsion is a kind of colloid. The term emulsion refers to a mixture in which the dispersed phase and the dispersion medium are both liquids. Demulsifiers, or emulsion breakers, are a class of specialty chemicals used to separate emulsions, for example, water in oil. Emulsion is a combination of two or more liquids that are ordinarily incompatible.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
